Stokes’ Law
What to do:
Push any of the four green buttons located around the bottom portion of the tube. Observe what happens. There are four different lasers and each one is a different color. Why do some lasers reach the end of the tube and others do not?
The red laser (wavelength of 650nm ± 10nm) passes through all four different colored discs and is seen on the end cap of the tube. This is because red light has very low energy and is not able to excite any of the electrons in any of the discs.
The green laser (wavelength of 532nm ± 10nm) is seen passing through the red disc and making it most of the way through the yellow disc. This is because, unlike the red light, the green light has enough energy to excite the electrons in both of the first two discs. The green light needs most of its energy in order to excite the electrons in the yellow disc and is therefore absorbed quickly. If the green light were able to make it to the green or blue discs, it would simply pass though, much like the red laser, because it does not have enough energy to excite the electrons in those discs.
The blue laser (wavelength of 405nm ± 10nm) is seen passing though the first three discs and then making it about a quarter of the way though the blue disc. This is because it has enough energy to excite the electrons in all four discs. However, it takes most of its energy to excite the electrons in the blue disc and is therefore rapidly absorbed.
The blue/UV laser (wavelength of 405nm) is seen passing though all of the discs and can be seen on the end cap of the tube as well. This light has so much energy that it is able to excite the electrons in all four of the discs and still have enough energy to make it to the end of the tube.
Reason:
Stokes’ Law states that when light hits an electron that is orbiting an atom, the electron can absorb that light and go into an excited state. In order for the electron to absorb the light, however, the light must have enough energy. Energy of light is dependent on its wavelength, or color. The shorter the wavelength, or the closer the color of light is to blue or ultra violet, the more energy the light has (see image below). This excited state for the electron does not last very long and the electron then goes back to its normal state.
When the electron goes from an excited state back to normal it releases the energy it received from the light. The energy released is also light and is sent off in a random direction (this is the reason you are able to see the light inside the discs). This new light, however (and this is the most important part), has less energy than the original light. Notice the colors of the four discs. The reason you are seeing these different colors is because of the fluorescent dyes in them. More importantly, the fluorescent dyes in these discs follow Stokes’ law.
Unlike normal objects, which either filter or absorb every color of visible light except the one you see, fluorescent dyes create their light. These dyes, due to their chemical make up, absorb all light that shines onto them whose energy is higher than the color of the disc. The dyes then release this energy as light of the color that you are seeing. This means that if you had two objects of the same color but one had fluorescent dye while the other had normal dye, the fluorescent object would be brighter. This is because the normal dyed object can only reflect the amount of color the room light shines on it, while the fluorescent dye will reflect that color and convert all higher energy light to that same color. These fluorescent dyes are called dayglo. Any light that shines on the discs that has the same energy or less than the light the disc is shining out, simply passes through without getting absorbed. This is seen with the red laser.
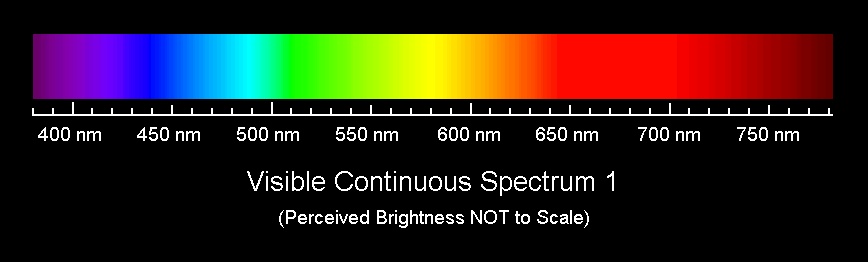
Shorter wavelength
Longer wavelength