Once only a part of science fiction, lasers are now everyday objects used in research, healthcare and even just for fun. Previously available only in low-energy light, lasers are now available in wavelengths from microwaves through X-rays, opening up a range of different downstream applications.


In a new study published online June 11 in the journal Nature, an international collaboration led by scientists at the University of Wisconsin–Madison has generated the shortest hard X-ray pulses to date through the first demonstration of strong lasing phenomena. The resulting pulses can lead to several potential applications, from quantum X-ray optics to visualizing electron motion inside molecules.
“We have observed strong lasing phenomena in inner-shell X-ray lasing and been able to simulate and calculate how it evolves,” says Uwe Bergmann, physics professor at UW–Madison, and senior author on the study. “When you calculate the X-ray pulses that come out, they can be incredibly short — shorter than 100 attoseconds.”
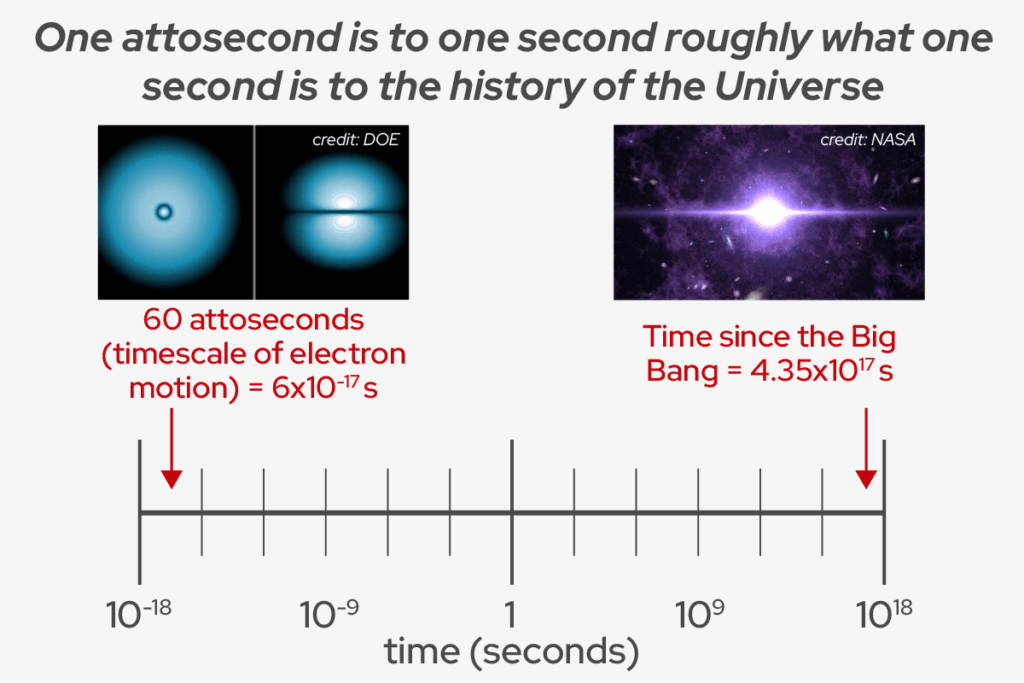
An attosecond is one quintillionth of a second and this extremely short duration of the pulses is what could drive new, advanced LASER applications.
The inner-shell X-ray lasing process is similar as in optical lasing, just at a much shorter wavelengths. An initial pulse of X-ray photons excites atoms’ inner-shell electrons. These excited electrons then emit photons of different X-ray wavelengths as they return to their state. Their emitted photons sometimes hit an already-excited atom, leading to an avalanche of stimulated emission radiation (the SER of LASER) in one direction.
Because inner-shell electrons are held very tightly, powerful X-ray pulses, like those from X-ray free-electron lasers (XFEL), are required to excite enough of them simultaneously to result in lasing. In turn, the photons they emit in this process are also at X-ray wavelengths. But XFEL pulses are generally “dirty,” with each pulse really being made of several short, intense spikes in time, and a range of spikes with different wavelengths, limiting some of their applications.
“They’re just not clean, beautiful pulses (like visible lasers),” says Thomas Linker, joint postdoctoral researcher at UW–Madison and the Stanford PULSE Institute at SLAC and lead author of the publication. “But it’s the only thing we have. We have to live with it.”
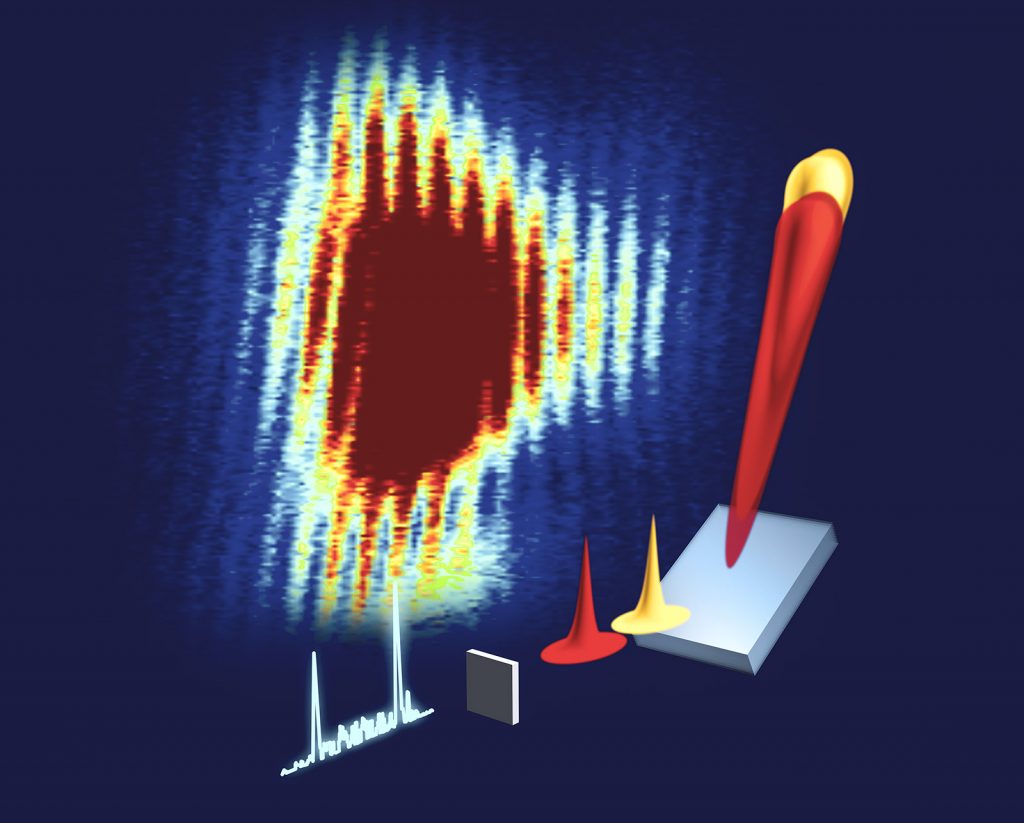
In this study, the researchers tightly focused XFEL pulses onto a sample made of copper or manganese. The input pulse is still dirty, but very short and incredibly powerful: the equivalent of focusing all the sunlight that hits the Earth into one square millimeter. The X-ray photons that the sample emits in the same forward direction as the input pulse hit a piece of instrumentation that disperses them by wavelength, much like a prism disperses visible light into a rainbow, and reflects it based on its angle. This dispersed X-ray light is next read by a detector, which measures its properties.
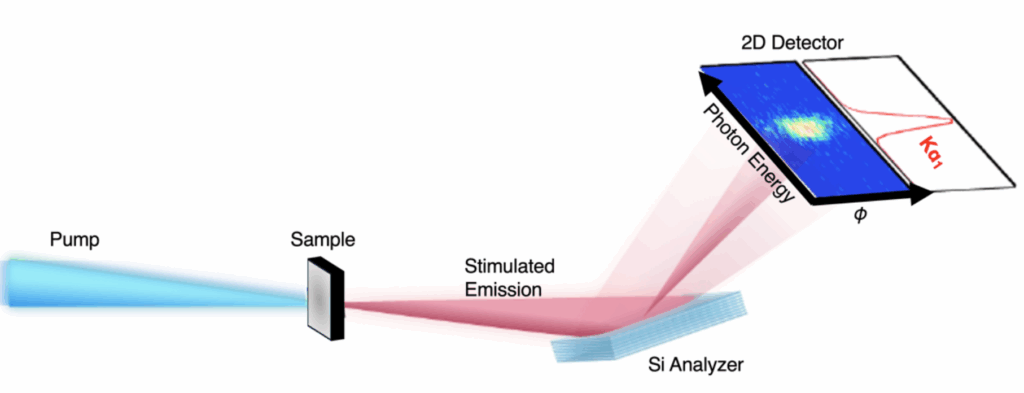
First, the researchers confirmed that stimulated emission is occurring in their sample by measuring a strong signal in the detector.
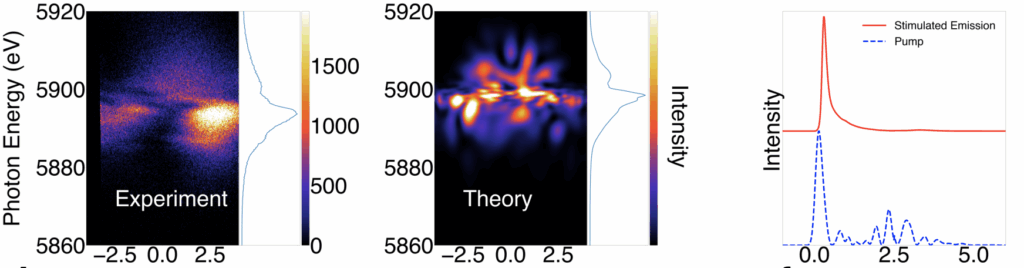
They noticed something else about the emitted light. In terms of its light spectrum, it contained all of the expected wavelengths. Spatially, however, the team sometimes detected a few hotspots instead of the expected smooth signal. Applying a 3D simulation, Linker was able to show what was happening to lead to these results. His calculations illustrated that the emitted X-rays underwent a process that created filaments when moving through the samples.
“This is filamentation, a strong lasing phenomenon which, in optical science, is when the index of refraction changes because of this very, very strong field,” Linker says. “You get spatial phenomena leading to the observed hotspots.”
When the team further increased the intensity of their input pulse, they saw another unexpected result: instead of seeing hotspots of one wavelength, they observed spectral broadening and sometimes multiple spectral lines. They ran the simulation on this new data and realized that this result can only be explained by another lasing phenomenon called Rabi cycling, where the pulse is so strong that the sample will cyclically absorb photons and emit them by stimulated emission. They used their simulation to plot the emitted pulse intensity over time and found that their dirty input pulses resulted in extremely short stimulated emission pulses that were sometimes 60-100 attoseconds in length — the shortest hard X-ray pulses observed by anyone to date.
“We have generated hard X-ray attosecond pulses with this strong lasing phenomenon,” Linker says. “The timescale at which chemical bonds are formed and broken is the femtosecond (1,000 times longer than attosecond) timescale. But if you want to see electron dynamics, how they move inside their orbitals, that’s the attosecond timescale.”
XFELs have only been around for about 15 years, so scientists are still learning about them and how to apply them. This study is not the first to “clean up” hard X-ray pulses, but it is the first to achieve emitted pulses on this timescale and to show evidence of strong lasing phenomenon.
“There are so many nonlinear technologies and phenomena that the laser community uses now, but very few of those have dared to have been tried with hard X-rays,” Bergmann says. “Hard X-rays are very powerful: they have Angstrom wavelengths that provide atomic spatial resolution, and they are sensitive to different elements. This work is a step towards pushing the exciting field of real laser science into this powerful hard X-ray regime.”
This work was largely supported by the U.S. Department of Energy (DOE), Office of Science, Basic Energy Sciences (BES) under contract Nos. DE-SC0023270 and DEAC02-76SF00515. The experiments were performed at the Linac Coherent Light Source at SLAC National Accelerator Laboratory and the SACLA X-ray laser at the Japan Synchrotron Radiation Research Institute